Whistle Use in Agonistic Interactions
of Bottlenose Dolphins
by Frank Veit

Introduction
Marine mammals are well known for their highly advanced vocal system, and a special part of it is the system of ‘signature whistles’ used, for example, by bottlenose dolphins (Tursiops truncatus). ‘Signature whistles’ are individual-specific signals, produced with a high degree of stereotypy, and their proposed function is to serve individual recognition and group cohesion (Janik & Slater 1998). In situations of isolation, they are the almost exclusively produced whistle vocalisations (review in Caldwell et al., 1990). Beside this signal system, bottlenose dolphins produce many other types of whistles, especially when socially interacting. However, as opposed to ‘signature whistles’, no associations between specific whistles and specific contexts could be shown so far. In this paper, I will report on the use of non-signature whistles. I will concentrate on the occurrence of specific whistles that were produced in the context of agonistic chases within a group of spontaneously interacting bottlenose dolphins.
Methods
Data on the occurrence of whistles in agonistic interactions were collected in summer 1994 in a period of 15 consecutive weeks. Site of the study was the Dolphin Reef (Eilat, Israel), a semi-free confinement in the Gulf of Aqaba, Red Sea. A group of six bottlenose dolphins lived at the site, supplemented by two calves born during the observation period. The animals, including adults of both sex (2,3), were maintained without separation of single individuals or subgroups. That means, also in periods like the mating or birthing season, all the animals stayed together, allowing the development of a complex social network. Recordings were made with two hydrophone systems (B&K 8103 hydrophones, placed 27 m apart, and B&K 2635 preamplifiers), connected to a video camera (Sony CCD-V6000E).
The occurrence of three non-signature whistle types, were analysed for the context of agonistic chases between adult individuals. The main characteristic of agonistic chases was that two or more animals were swimming rapidly, one (actor) behind the other (recipient) and without changes of roles within one episode. The interaction could include physical attacks like bites or body slams. Additionally, different sub-contexts are compared, defined by the involved individuals and their respective role, either pursuing or being pursued. Analysed were whistle vocalisations from the onset of the interaction until one minute after its end. As a control, 45 randomly selected samples of 20 minutes (= 900 min), during which no agonistic behaviours occurred, were analysed for the occurrence of the three whistle types. The control samples covered a broad spectrum of conditions, like play, foraging, or resting. Categorisation of the whistle vocalisations was done by visual inspection of the spectrograms.

Results
Whistles
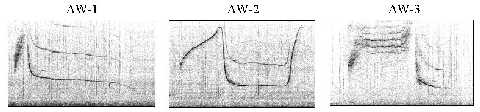
Non-signature whistles were frequently produced during agonistic interactions. Figure 1 shows spectrograms for the three types under investigation, termed AW-1, AW-2, and AW-3. They were discrete types, that is, parametric variation within the types was much smaller than between them. A common feature of these whistles, and unusual for whistles of the species, was, that they were produced almost exclusively as single loops. That means, repetitive loops were separated by silent intervals. The total number of the three whistle types in the sample was 244.
Figure 1. Examples of the three agonistic whistle types.
Contexts
Of the five adult dolphins of the group, four were involved in chases: the two males (termed MALE-1 [Cindy] and MALE-2 [Dicky]) and two of the females [Domino and Dana]. A very close association existed between these two females, which gave birth within a few hours that summer. They synchronised their activities during most times of the day for the whole data sampling period. This was also true in chasing episodes. When, for instance, one was chased by a male, the second female often swam at her side. Therefore it was often impossible to decide to whom of the two the aggression was directed, and I did not differentiate between them but treated them as one unit (termed FEMALES). MALE-1 and MALE-2 both acted as pursuers in this dyad. The FEMALES were only the recipients of chases, and only MALE-1 was pursuing them.
Occurrence of Whistles
As a first step, the number of chasing episodes and control samples are compared, in which at least one of the three whistle types were produced at all. In only three of the 45 control samples, one of the whistles occurred (always one of type AW-1). Non of the whistles occurred in any of the eleven chasing episodes of MALE-2 behind MALE-1. The majority of all the three whistle types occurred, when MALE-1 was the actor. They were produced in 87.5 % of the chases behind the FEMALES, and in 62.5 % of the chases behind MALE-2. The differences in the occurrence of the whistles (AW-1, -2, -3) for the compared contexts were significant (c2 test for ‘goodness-of-fit’, c2>200, P<0.01, 1 df). These trends would become even more evident, when the total numbers of the whistles are compared for the contexts.
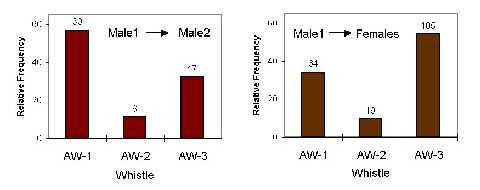
Figure 2. Relative frequency of the three AW-types in the two contexts, where Male1 was the pursuer. The total number of whistles is given above bars.
Altogether 241 of the specific whistles occurred, when MALE-1 was the actor: 53 when he was pursuing MALE-2, and 188 when he was pursuing the FEMALES. Fig. 2 shows, how the occurrence of the three whistle types differs for these two contexts. A significant difference existed in the production of the whistles for the two contexts (c2 = 10.071, P<0.01, 2 df). When MALE-1 was pursuing MALE-2, the AW-1 type was produced significantly more frequent (binomial test, Bonferroni corrected, two-tailed, Z = 2.323, P<0.05), and when he was pursuing the FEMALES, it was the AW-3 type (Z = -2.149, P<0.05).
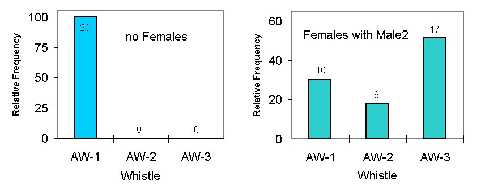
Before continuing with the results, another short excursion to the social relationships within the group has to be made. In many of the chasing episodes, other animals than the directly involved ones (actor and recipient) accompanied the interaction. This was, for example, the case in ten of the 24 chases of MALE-1 behind MALE-2, when one or both FEMALES joined MALE-2 during the interactions by swimming parallel to him, and forming a subgroup with him just after. In Fig. 3, again the relative frequency of the three whistle types is shown for the context of MALE-1 pursuing MALE-2 (left graph of Fig. 2), but now the context is splitted according to the behaviour of the FEMALES, that is swimming with MALE-2 or not. When no FEMALE joined in chases of MALE-1 behind MALE-2, only AW-1 types occurred (left graph of Fig. 3). When the FEMALES joined in the interaction, all three whistle types were produced. This means, that types AW-2 and AW-3 occurred exclusively, when the FEMALES were involved in a chasing episode, either directly pursued by MALE-1 (right graph of Fig. 2), or joining MALE-2 while he was pursued by MALE-1 (right graph of Fig. 3).
Figure 3. Relative frequency of the three AW-types for chases of Male1 behind Male2 according to the behaviour of the Females. The total number of whistles is given above bars.
Discussion
Three discrete whistle types were presented, which in the studied group were given in the context of chasing. They showed a degree of stereotypy comparable to ‘signature whistles’ of the species, and this stereotypy even counted for their loop number, as they were produced almost exclusively in single loops. Yet repetitions of the signals did happen, as the whistles were often produced in series. All three whistles were closely associated to agonistic contexts, and are therefore labelled ‘agonistic whistles’ (AW). The occurrence of specific whistles (types AW-1, -2, -3) was related to details of the context of chasing, for example the presence of females (Fig. 2 and 3). These close associations between whistle types and social contexts suggests that they indeed serve a particular communicative function.

Recently, few types of low-frequency sounds were described for both, wild and captive bottlenose dolphins, which showed a close correlation to specific social contexts. Connor et al. (1996), for example, described “pops” produced by males during consortship behaviour in a wild population, and McCowan and Reiss (1995) interpreted “thunks” emitted by females in one captive group as an aggressive contact vocalisation towards infants. As mentioned above, no such associations were found for whistle vocalisations previously - excluding the so-called “distress whistles” described by Lilly (1963), which were later interpreted as the probable ‘signature whistles’ of the investigated animals, likely to be produced in distressing situations (Herman & Tavolga, 1980).
The results of this paper suggests that, for a better understanding of the communication system of bottlenose dolphins, more attention should be paid to the big group of signals that are labelled here as non-signature whistles. The three AW-types are the first non-signature whistles of the species, for which a contextual production could be demonstrated conclusively.
REFERENCES:
-
Caldwell, M. C., Caldwell, D. K. and Tyack, P. L. 1990. Review of the signature-whistle hypothesis for the Atlantic bottlenose dolphin. Pp. 199-234. In The bottlenose dolphin. (Eds. S. Leatherwood and R.R. Reeves). Academic Press, San Diego. 653pp.
-
Connor, R. C. and Smolker, R. A. 1996. ‘Pop’ goes the dolphin: a vocalization male bottlenose dolphins produce during consortship. Behaviour, 133: 643-662.
-
Herman, L. M. and Tavolga, W. N. 1980. The communication system of cetaceans. Pp. 149-209. In Cetecean behavior: mechanisms and functions. (Ed. L. M. Herman). Wiley, New York. 463pp.
-
Janik, V. M. & Slater, P. J. B. 1998. Context-specific use suggests that bottlenose dolphin signature whistles are cohesion calls. Animal Behaviour, 56, 829-838.Lilly, J. C. 1963. Distress call of the bottlenose dolphin: stimuli and evoked behavioral responses. Science, 139: 116-118.
-
Lilly, J. C. 1963. Distress call of the bottlenose dolphin: stimuli and evoked behavioral responses. Science, 139, 116-118.
-
McCowen, B. and Reiss, D. 1995. Maternal aggressive contact vocalizations in captive bottlenose dolphins (Tursiops truncatus): wide band, low frequency signals during mother/aunt-infant interactions. Zoo Biol., 14: 293-309.
Source:
-
Presentation at the 1st World Marine Mammal Conference, Monaco, 1998.